
NKI-AvL
Dr. Marleen Kok (NKI-AvL)
Prof. dr. Gabe Sonke (NKI-AvL)
Prof. dr. Agnes Jager (Erasmus MC)
Femke Verwer (Clinical Projects Manager NKI-AvL)
E: planet@nki.nl
Dr. Elise van Leeuwen-Stok (Clinical Study Manager BOOG Study Center)
Ilse Schilderinck, MSc (project assistant BOOG Study Center)
E: info@boogstudycenter.nl
T: 088-234 6730
Dr. Annemiek van Ommen-Nijhof (NKI-AvL)
Drs. Robin van den Borg (NKI-AvL)
E: planet@nki.nl
NKI-AvL
NKI-AvL
Centrum zelf of IKNL
ZE&GG / Ministry of Health
Stichting Treatmeds
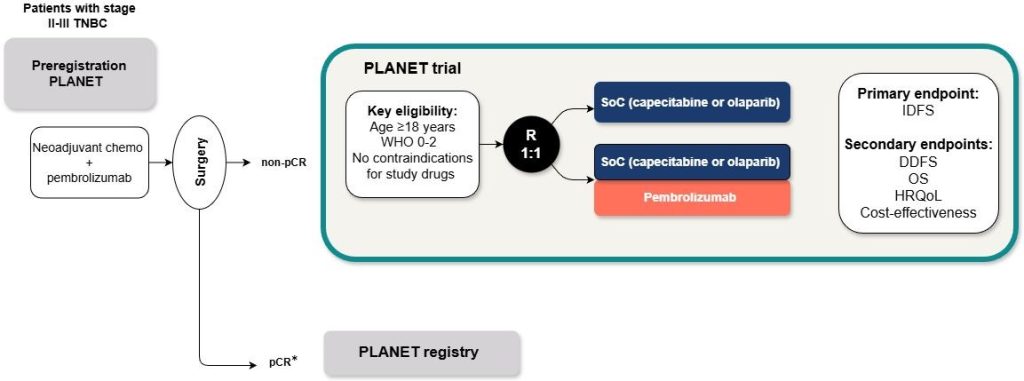
The PLANET trial is a randomized controlled phase 3 trial with two arms and a superiority design. Randomization will be stratified according to initial clinical disease stage (stage II versus III), residual tumor size (≥1cm or <1cm), prior platinum exposure and intended standard of care treatment (capecitabine versus olaparib).
Only participants with non-pCR are eligible for participation in the PLANET trial (see Chapter 7 of the protocol on detailed eligibility criteria); participants with pCR after neoadjuvant treatment will be followed through the PLANET pCR registry.
|
|
Primary objective:
To evaluate the value of adding adjuvant pembrolizumab to standard adjuvant treatment in patients with TNBC who have residual disease after neoadjuvant chemotherapy plus pembrolizumab
Secondary objectives:
Primary endpoint:
The primary endpoint of the study will be invasive disease-free survival (IDFS), defined as time since randomization to local or distant breast cancer recurrence, second primary non-breast cancer or death due to any cause, whichever occurs first. Patients without any of these events will be censored at the time of the analysis.
Secondary endpoints include:
Main inclusion criteria:
Exclusion criteria: